
Pan-London guideline for in utero transfer
| Developed by: | London Maternity Clinical Network and London Neonatal Operational Delivery Network | |
| First Published: | September 2018 | |
| Review date: | August 2019 | |
| Contact: | england.maternityscn@nhs.net | |
| Date signed off: | 23 August 2018 NHS England (London) Joint Quality Committee |
21 August 2018 NHS England (London) Maternity Clinical Leadership Group |
1. Purpose
In utero transfers are required when pregnant women are likely to deliver a baby who requires expertise or not available at the local unit; or when pregnant women have illness in pregnancy requiring expertise not available at the local maternity unit. Ex utero transfers (postnatal transfers) that occur immediately after birth are associated with increased neonatal morbidity and mortality and some of these represent missed opportunities for in utero transfers and some of these represent missed opportunities for in utero transfers.
The guideline has been produced by the pan-London In Utero Transfer working party hosted, by the London Maternity Clinical Network following consultations with the Emergency Bed Service (EBS) at the London Ambulance Service (LAS). It is intended to build upon the British Association of Perinatal Medicine (BAPM) 2008 document on management of acute in utero transfers.
This guidance aims to achieve the following:
- Ensure timely in utero transfers for those pregnant mothers most at risk.
- Reduce the number of unnecessary transfers.
- Improve the patient experience of transfer.
- Decrease the time spent arranging transfer by clinicians.
The guideline does not aim to replace local network or trust clinical guidelines for in utero transfer. The guideline provides information on key areas of the in utero pathway and will help inform both the content and structure of local guidelines. The goal of the guideline is to better inform clinicians, support their decision-making process and reduce inappropriate variation in practice.
2. Introduction and background
The inefficiencies of arranging in utero transfers in the London region for expectant mothers are well known among maternity and neonatal staff. The overall success rate of arranging transfers was 63 per cent in 2012, according EBS data. Of successful transfers, 33 per cent were transferred to units not recommended by EBS. Analysis of EBS referrals for in utero transfer revealed a median duration of 340 minutes for a successful transfer. A 2013 survey of obstetric and neonatal doctors confirmed the dissatisfaction with the current system and an appetite for new processes, increased transparency and consensus on standards.
The issues militating against effective in utero transfers include:
- Failure to recognise the need for higher level neonatal care in extremely preterm babies and the improved outcomes that result from the care being delivered in neonatal intensive care units (NICUs).
- Difficulty in predicting preterm birth, which can result in both the failure to transfer pregnant women in a timely way and the transfer of pregnant women who do not go on to deliver after transfer.
- Insufficient neonatal intensive care capacity.
- Insufficient capacity in maternity units.
- Lack of real time information, such as presence of available neonatal capacity.
- Lack of national guidance on best practice related to referral and acceptance process between hospitals. This would include organising maternal transfer.
Being transferred out of local area is associated with a significant stress and expense for the pregnant woman and her family. There is an immediate clinical burden of transfer arrangements3, and a midwife may be required to accompany the pregnant woman during the transfer. Paradoxically, unwarranted antenatal transfers may increase more dangerous postnatal transfers by impairing efficient management of neonatal cots (cots are reserved for babies that are not born, reducing access for babies needing postnatal transfer). With these neonatal cots occupied, babies of pregnant women in true preterm labour are only able to be transferred postnatally.
Neonatal care services are provided in a variety of settings, dependent upon the interventions required for the baby. Neonatal services are organised across three levels of care according to expertise and staffing levels, so that not all neonatal units (NNUs) are commissioned to care for all babies. Details on categories and levels of neonatal care are provided in appendices A and B. Designations of London NNUs are provided in appendix C.
3. Identification and assessment of evidence
The Cochrane Library (including the Cochrane Database of Systematic Reviews), DARE, Embase, TRIP, Medline and PubMed (electronic databases) were searched for relevant randomised controlled trials, systematic reviews and meta-analyses and cohort studies. The search was restricted to articles published during 2006 to 2016. Search words included ‘in utero transfer’, ‘neonatal transport’ and ‘ex utero transfer’. The search was limited to humans and to the English language. All relevant articles identified are listed in section seven.
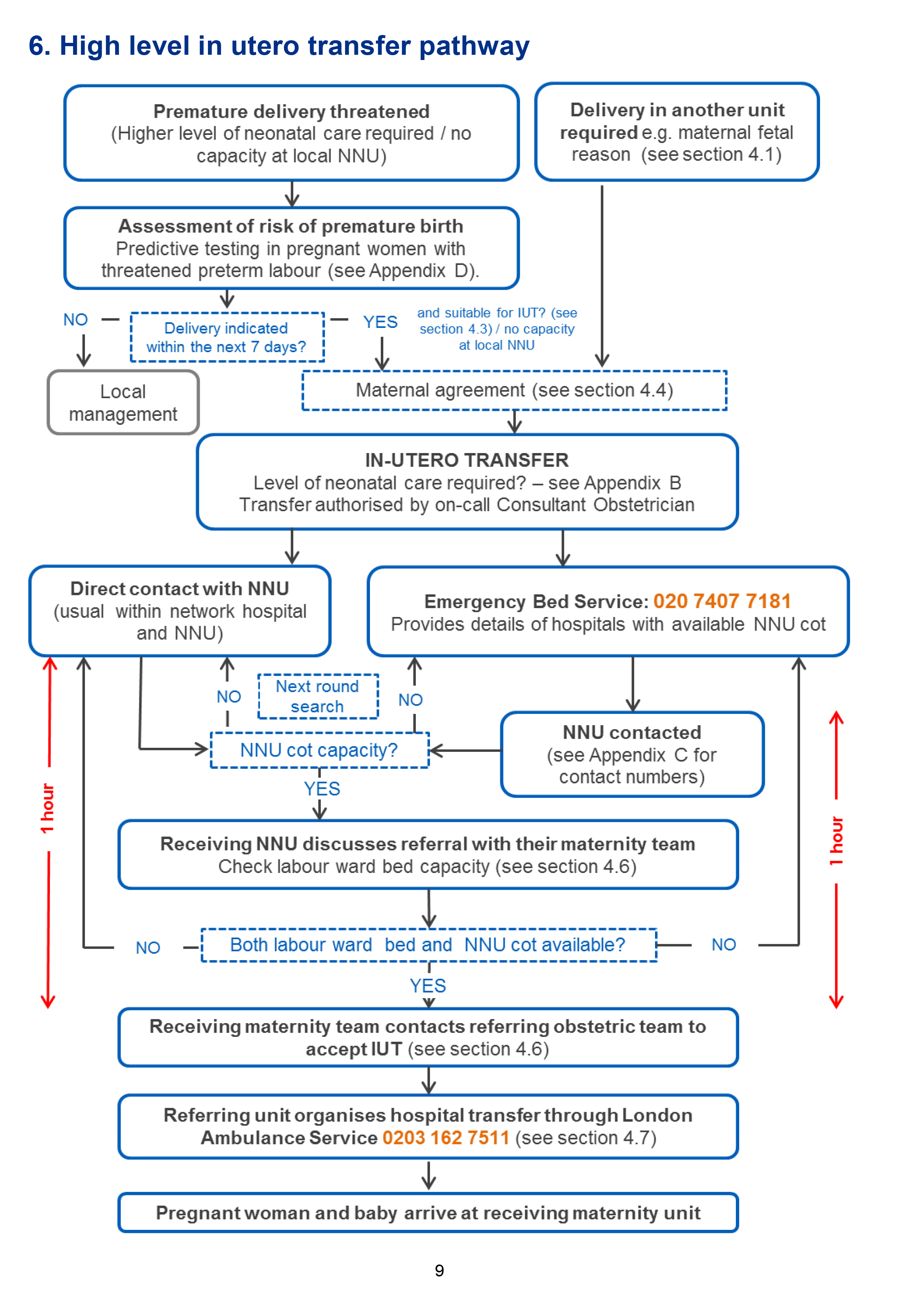
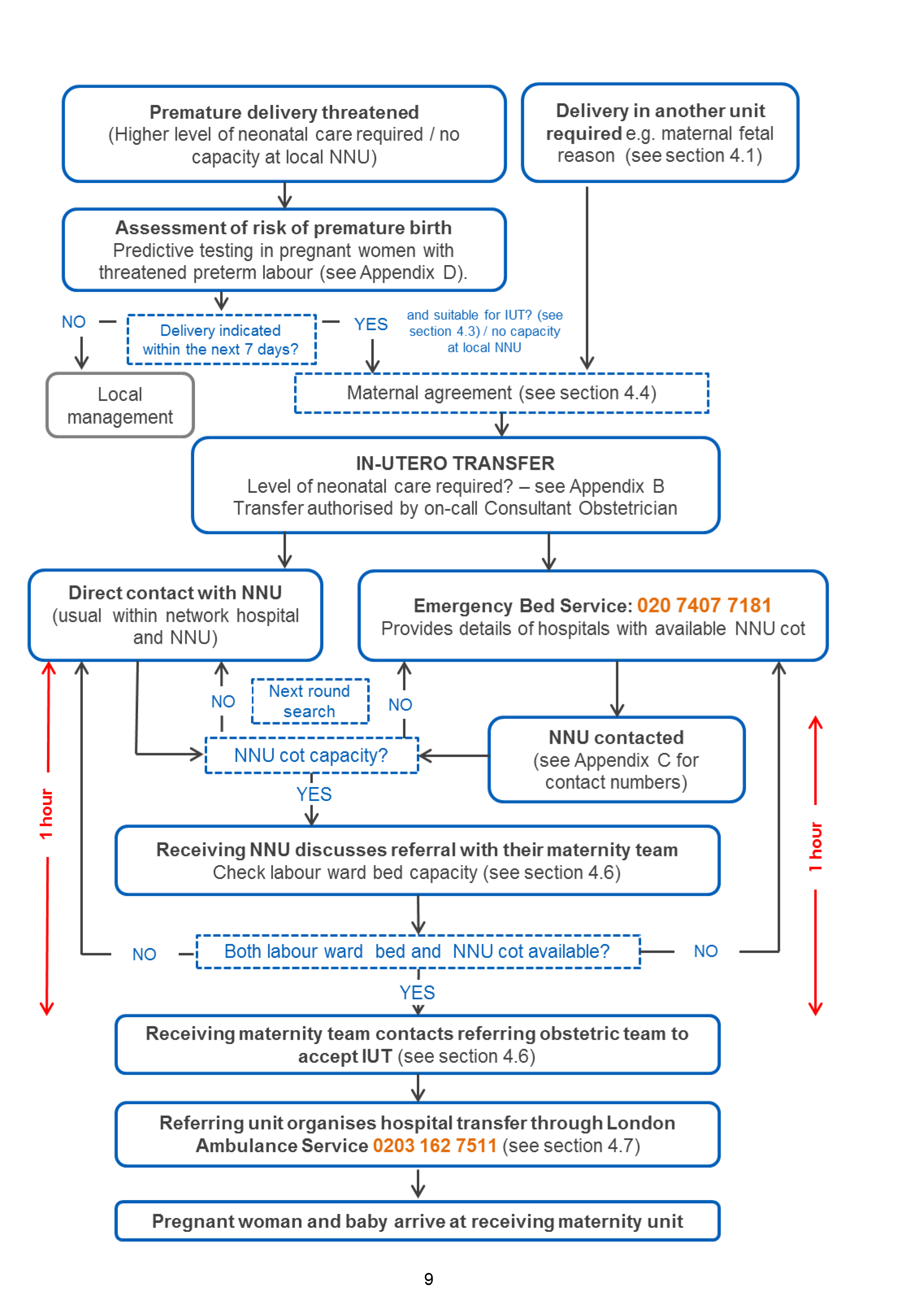
4. Arranging in utero transfer
4.1 When is in utero transfer indicated?
An in utero transfer is indicated when there is an increased risk of delivery within the next seven days for one or more of the following reasons.
- Maternal medical or surgical condition needing tertiary level input.
- Fetal medical or surgical condition needing iatrogenic delivery at tertiary centre with facilities and capacity for neonatal management, including for any diagnosed subspecialty condition (cardiac).
- High risk of spontaneous or iatrogenic birth in a unit without facility or capacity to manage the newborn (usually due to prematurity) as per national service specification. Refer to appendices B and C.
For pregnant women who are suspected to be in preterm labour, the risk of premature birth should be assessed with, at least, quantitative fetal fibronectin and/or cervical length measurements before a decision to transfer is made. Refer to appendix D.
Precise thresholds for intervention will depend on the clinical setting, but it is recommended that a risk of delivery of less than 5 per cent in the next week may justify withholding antenatal steroid administration, admission or in utero transfer.
4.2 Which patients are not suitable for in utero transfer?
The following patients are not normally considered suitable for in utero transfer:
- Pregnancy less than 23 weeks for fetal reasons – Lower or uncertain gestations could be considered on a case by case basis. In some circumstances it is appropriate to transfer a pregnant woman at gestation between 22 and 23 weeks.
- Potentially lethal fetal condition where active intervention of the fetus was not being considered even if live born. (In cases of fetal abnormalities it is useful to discuss these cases with fetal medicine specialists.)
- Active labour where the cervix is more than three centimetres dilated.
- Maternal condition which may require intervention during transfer (antepartum haemorrhage or uncontrolled hypertension).
- Known maternal or fetal compromise requiring immediate delivery, including abnormal Cardiotocography (CTG).
These exclusion criteria have been adapted from the BAPM 2008 document, ‘Management of acute in utero transfers’.
4.3 Who makes the decision for in utero transfer?
All potential transfers must be authorised by the on call obstetric consultant, following discussion with the consultant neonatologist/paediatrician of the referring hospital.
The aim of consultant involvement is to ensure that the assessment of risk is appropriate and the resource implications and implications for the pregnant woman and baby are taken into account before an in utero transfer is embarked upon
4.4 What about maternal agreement to be transferred?
Maternal agreement needs to be obtained prior to transfer. This should be documented in the maternal healthcare record. This should involve counselling by the obstetric and neonatal staff.
The distress and inconvenience of in utero transfer needs to be recognised and its clinical indication explained to the patient, including the lack of local facilities for higher levels of neonatal care and implications for neonatal outcome. Possible transfer of the baby (or babies) back to their original hospital, or one nearer home, after completion of intensive and/or high dependency neonatal care should also be explained.
4.5 Finding a maternity bed and a neonatal cot – current process
Once the need for in utero transfer is identified, a local neonatal unit (LNU) or NICU (depending on the baby and likely level of care required) and an associated maternity unit will need to be found to accept the transfer.
In London, there are currently two options available:
- Referring unit will directly contact their usual network hospital neonatal unit. See appendix C for contact telephone numbers.
- Referring unit will contact the EBS to obtain a cot status of appropriate neonatal units.
EBS gathers neonatal cot availability twice daily by phoning every neonatal unit in London, Kent, Surrey and Sussex.
Referring hospitals who contact EBS will obtain a list of hospitals with cots available, in order of most appropriate referral pathways, to allow prioritisation of calls to referring hospitals.
Referring units should recognise that a search for a specific space for a pregnant woman will always require a direct approach and discussion. Cot status should provide a general view of availability, against which a more focussed search can take place.
It is recognised that some neonatal units are inputting their data into a tool, Cot Finder app, to share the number of cots available in their unit. The app is designed to help maternity staff identify cot availability across London, allowing a more focused search to take place.
4.5.1 Future development
The London Maternity Clinical Network and London Neonatal Operational Delivery Network are pursuing improving the cot finding element of the in utero pathway. This will include developing a centralised timely system/process for locating the required level of neonatal cot within hospitals and effectively managing timely transfers of mother and baby in utero.
A process is in development for sharing red, amber, green (RAG) status data via a content management system which provides information on maternity bed status. Work is ongoing to develop electronic processes to collect and share this data. It is likely the two developments will become interlinked.
4.6 Communication between referring obstetric team and receiving unit
One of the aims of this guidance is to ensure referring units do not have an undue burden of phone calls to successfully find a receiving unit and that decisions are made in a timely manner.
- The referring obstetric team will contact the potential receiving neonatal unit, identified through EBS or other method, to discuss with the neonatal team.
- On confirming the presence of neonatal capacity, the receiving neonatal team will discuss the referral with their maternity team (consultant obstetrician and labour ward coordinator who will have an overview of current labour ward capacity and activity).
- When the receiving unit decides that there is both neonatal and maternity capacity, the receiving maternity team will then contact the referring maternity unit to accept the transfer.
- Prior to transfer, the referring obstetric team should speak directly to the most senior obstetrician on site at the receiving hospital to discuss all relevant clinical details. In some circumstances, the maternal transfer should be at consultant to consultant level (extreme prematurity, severe maternal disease).
It is the responsibility of the receiving team to agree or refuse transfer within one hour of referral.
4.6.1 Acceptance of transfer
There is an expectation that a receiving hospital with NICU will accept an appropriate in utero referral from a Special Care Unit (SCU) or local neonatal unit within their network unless there are exceptional circumstances. These pregnant women will be considered similar priority to local pregnant women and pregnant women already on the labour ward.
There should be recognition by the receiving neonatal unit that unless delivery is likely to occur imminently after transfer, there will not necessarily need to be a physically vacant neonatal cot to accept the in utero transfer.
There should be recognition by the receiving maternity unit that (i) they should consider both antenatal ward and labour ward capacity in order to accept the transfer, as many pregnant women will be able to be managed on the antenatal ward after transfer and (ii) the immediate acuity of the labour ward may not impact on the ability to accept the referral, as the transfer is likely to take two or more hours to complete.
4.6.2 Refusal of transfer
A decision of the potential receiving neonatal team to refuse acceptance of an appropriate transfer should be made only after consultation with the neonatal consultant in charge.
A decision of the potential receiving maternity unit to refuse transfer should only be made after consultation between the midwife in charge of the labour ward and the obstetric consultant on duty or on call.
4.7 Ambulance transfer
London Ambulance Service (LAS) telephone number for hospital transfer: 0203 162 7511.
- Once in utero transfer has been accepted by the receiving neonatal and maternity unit, the referring unit should organise the transfer through LAS. Requests to LAS for a hospital transfer must be made by staff able to give appropriate clinical information and answer clinical questions asked by LAS.
- In utero transfer will generally be organised using LAS Hospital Transfer 3 priority, which provides a 120 minute response time.
- Provision of an escort from the referring maternity team for the transfer will be made on a case by case basis. This should be decided by the senior maternity staff on duty at the referring hospital, bearing in mind that LAS cannot guarantee the presence of a paramedic team for transfer.
- Photocopies of the relevant notes and investigations should accompany the mother for transfer. Hard copies will be retained in the referring hospital clinical notes. Where the network has an agreed ‘in utero transfer record’ document, this should be completed in all instances of attempted in utero transfer. A copy of this should be kept with the hospital clinical notes and a copy sent to local maternity clinical governance leads.
The emotional needs of the parents must be recognised. A birthing partner should be allowed in the ambulance unless clinically unsafe or their presence would compromise the care being given.
5. Audit and Oversight
All clinical involvement with any attempted in utero transfers should be recorded and available for clinical audit.
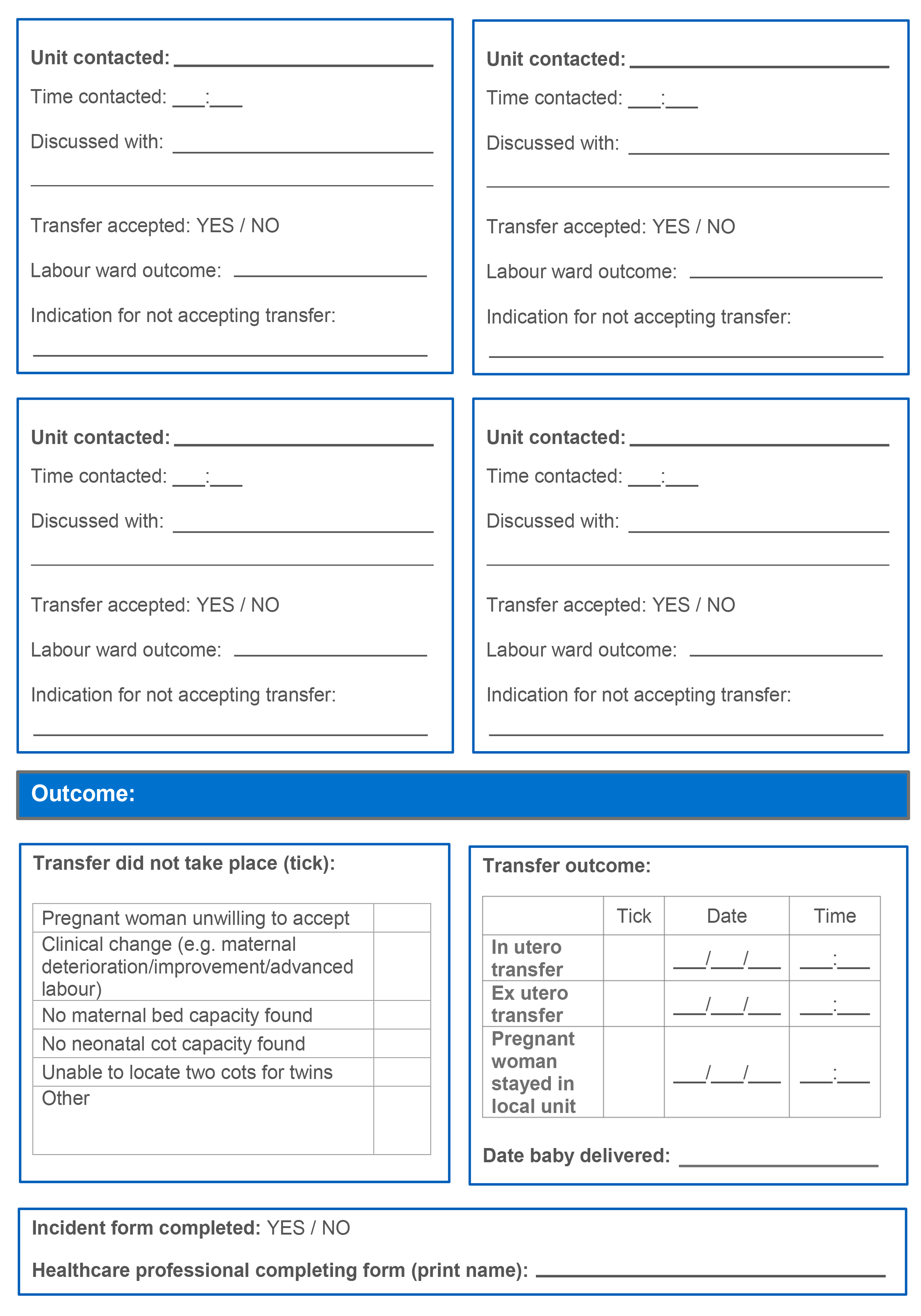
It is recommended that all Local Maternity Systems (LMSs) have an agreed ‘in utero transfer record’ document which should be completed in all instances of attempted transfer. A copy should be kept in hospital clinical notes and a copy sent to local maternity clinical governance leads. An example of this document can be found in appendix E.
Local Maternity Systems and Neonatal Operational Delivery Network (ODN) sectors are responsible for monitoring in utero and ex utero transfers. For example by audit and exception report monitoring with a particular focus on learning from missed opportunities for in utero transfer. As a minimum, Local Maternity Systems should audit the number of admitted babies born at less than 27 weeks of gestation delivered in maternity services that are not on the same site as a designated neonatal intensive care unit.
6. High level in utero transfer pathway
7. References
- Abbott, Danielle S, Samara K Radford, Paul T Seed, Rachel M Tribe, and Andrew H Shennan. "Evaluation of a Quantitative Fetal Fibronectin Test for Spontaneous Preterm Birth in Symptomatic Women." American journal of obstetrics and gynecology 208, no. 2 (2013): 122-e1.
- Fenton, A, D Peebles, and J Ahluwalia. "Management of Acute in Utero Transfers: A Framework for Practice." Report for the British Association of Perinatal Medicine (2008). [Online] Available at https://www.bapm.org/sites/default/files/files/IUTs_Jun08_final.pdf
- Gale, C, A Hay, C Philipp, R Khan, S Santhakumaran, and N Ratnavel. "In-utero Transfer Is Too Difficult: Results From a Prospective Study." Early human development 88, no. 3 (2012): 147-150.
- Mclaren J., Watson H., Survey of Current n-utero transfer processes in England Poster presented4 RCOG World Congress, India, March 2014.
- National Collaborating Centre for Women's and Children's Health (UK). Preterm Labour and Birth. National Institute for Health and Care Excellence: Clinical Guidelines. London: National Institute for Health and Care Excellence (UK), November, 2015. [Online} Available at https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0080792/
- Porcellato, L, G Masson, F O'Mahony, S Jenkinson, T Vanner, K Cheshire, and E Perkins. "‘It's Something You Have to Put Up With’—service Users’ Experiences of in Utero Transfer: A Qualitative Study." BJOG : an international journal of obstetrics and gynaecology 122, no. 13 (2015): doi:10.1111/1471-0528.13235. [Online] Available at:http://onlinelibrary.wiley.com/doi/10.1111/1471-0528.13235/full
8. Abbreviations
| EBS | Emergency Bed Service |
| LAS | London Ambulance Service |
| LMS | Local Maternity System |
| LNU | Local Neonatal Unit |
| NICU | Neonatal Intensive Care Unit |
| NNU | Neonatal Unit |
| ODN | Operational Delivery Network |
| SCU | Special Care Unit |
9. Appendices
9.1 Categories of neonatal care
Neonatal activity is linked to the following categories of care:
| Intensive care | Specialised care for the smallest and most seriously ill babies who require constant care and, often mechanical ventilation to keep them alive. Babies with severe respiratory disease and those who also require surgery will need this level of care too. |
|---|---|
| High dependency care | Care provided to babies who need continuous monitoring. For babies needing non-invasive breathing support, including receiving continuous positive airway pressure (CPAP). Babies receiving parenteral nutrition (intravenous feeding) also need this level of care. |
| Special care | The least intensive level of neonatal care and the most common. For babies who need continuous monitoring of their breathing or heart rate, additional oxygen, tube feeding, phototherapy (to treat neonatal jaundice) and convalescence from other care. |
| Transitional care | Babies who have special care needs but are able to be managed alongside the mother, who is the main carer; the mother is supported by neonatal staff alongside the midwifery team. In some services, transitional care occurs in the post-natal ward and, in others, in a discreet area or transitional care unit with staffing from both neonatal and midwifery teams. |
9.2 Levels of neonatal care
The Neonatal Critical Care service specification recognises three levels of neonatal unit, stratified by the level of neonatal care that they are commissioned and staffed to provide (https://www.england.nhs.uk/commissioning/wp-content/uploads/sites/12/2015/01/e08-serv-spec-neonatal-critical.pdf).
| Level 1 Special Care Unit (SCU) |
Commissioned and staffed to provide care for babies of births after 31+6 weeks gestational age provided the anticipated birth weight is above 1,000g (some ODNs have approved care pathway where babies born between 30+0 and 31+6 weeks gestational age receive initial care in Special Care Unit (SCU) provided the anticipated birth weight is above 1,000g and intensive care is not required. Some SCUs will only provide care for babies >33+6 weeks gestation). Not commissioned and therefore not be expected to provide:
|
|---|---|
| Level 2 Local Neonatal Unit (LNU) |
Commissioned and staffed to provide care for babies of singleton births after 26+6 weeks gestational age and multiple births after 27+6 weeks gestational age providing the anticipated birth weight is above 800g. Not commissioned and therefore not be expected to provide:
|
| Level 3 Neonatal Intensive Care Unit(NICU) |
Commissioned and staffed to provide care for all babies from birth, in line with national guidelines and professional standards, at all gestations after 22+6 weeks. All level 3 NICU service will also provide lower level neonatal support across their maternity catchment area. |
9.3 Designation of London neonatal units
Level 3: Neonatal intensive care units (NICUs)
| Chelsea and Westminster Hospital | Chelsea and Westminster Hospitals NHS Foundation Trust |
020 3315 7883 020 3315 7855 |
North West London ODN and LMS |
| Homerton Hospital | Homerton University Hospital NHS Foundation Trust |
020 8510 7364 / 7361 /7906 020 8510 7351 / 7352 |
North Central and East London ODN East London LMS |
| King’s College Hospital | King’s College Hospital NHS Foundation Trust |
020 3299 3553 020 3299 4700 |
South London ODN South East London LMS |
| Queen Charlotte’s and Chelsea Hospital | Imperial College Healthcare NHS Trust |
020 3313 3174 020 3313 4240 |
North West London ODN and LMS |
| The Royal London Hospital | Barts Health NHS Trust |
020 3594 0524 / 0511 020 3594 2367 /2369 |
North Central and East London ODN East London LMS |
| St George’s Hospital | St George’s University Hospitals NHS Foundation Trust |
020 8725 1936 020 8725 1921 |
South London ODN and South West London LMS |
| Evelina London Children’s Hospital | Guy’s and St Thomas’ NHS Foundation Trust |
020 7188 4045 020 7188 2233 /2973 |
South London ODN and South East London LMS |
| University College Hospital | University College London Hospitals NHS Foundation Trust |
020 3447 6233 020 7447 6203 /6204 |
North Central and East London ODN and North Central London LMS |
Level 2: Local neonatal units (LNUs)
| Barnet Hospital | Royal Free London NHS Foundation Trust |
020 8216 5160 / 5161 020 8216 5180 |
North Central and East London ODN and North Central London LMS |
| Croydon University Hospital | Croydon Health Services NHS Trust |
020 8401 3191 020 8401 3179 / 3180 |
South London ODN and South West London LMS |
| Hillingdon Hospital | The Hillingdon Hospitals NHS Foundation Trust |
01895 279 447 /109 01895 279 441 |
North West London ODN and LMS |
| Kingston Hospital | Kingston Hospital NHS Foundation Trust |
020 8546 2421 / 2420 020 8934 3117 / 3118 /2422 |
South London ODN and South West London LMS |
| Lewisham Hospital | Lewisham and Greenwich NHS Trust |
020 333 3140 / 3139 020 8333 3026 |
South London ODN and South East London LMS |
| Newham Hospital | Barts Health NHS Trust |
020 7363 8700 / 8138 020 7363 8737 / 8738 |
North Central and East London ODN and East London LMS |
| North Middlesex Hospital | North Middlesex University Hospital NHS Trust |
020 8887 2643 / 2966 020 8887 2500 |
North Central and East London ODN and North Central London LMS |
| Northwick Park Hospital | London North West Healthcare NHS Trust |
020 8869 2900 / 2902 020 8869 2890 |
North West London ODN and LMS |
| Queen Elizabeth Hospital | Lewisham and Greenwich NHS Trust |
020 8836 4530 / 4394 020 8836 4522 |
South London ODN and South East London LMS |
| Queen’s Hospital | Barking, Havering and Redbridge University Hospitals NHS Trust |
01708 435 115 01708 435 371 |
North Central and East London ODN and East London LMS |
| St Helier Hospital | Epsom and St Helier University Hospitals NHS Trust |
020 8296 2885 020 8296 2479 |
South London ODN and South West London LMS |
| St Mary’s Hospital | Imperial College Healthcare NHS Trust |
020 3312 6294 020 3312 1722 |
North West London ODN and LMS |
| Whipps Cross Hospital | Barts Health NHS Trust |
020 8535 6513 020 8535 6573 |
North Central and East London ODN and East London LMS |
| Whittington Hospital | Whittington Health NHS Trust |
020 7288 5530 020 7288 5502 |
North Central and East London ODN and East London LMS |
Level 1: Special care units (SCUs)
| Epsom Hospital | Epsom and St Helier University Hospitals NHS Trust |
01372 735 279 01372 735 208 |
South West London ODN and LMS |
| Princess Royal University Hospital | King’s College Hospital NHS Foundation Trust |
01689 864 956 / 957 01689 864 811 |
South London ODN and South East London LMS |
| Royal Free Hospital | Royal Free London NHS Foundation Trust |
020 7830 2733 020 7830 2721 |
North Central and East London ODN and North Central LMS |
| West Middlesex Hospital | Chelsea and Westminster Hospitals NHS Foundation Trust |
020 8321 5944 / 5945 020 8321 5946 / 5947 |
North West London ODN and LMS |
Acute hospitals in London with no obstetric unit or neonatal service
| Central Middlesex Hospital | London North West Healthcare NHS Trust | |
| Charing Cross Hospital | Imperial College Healthcare NHS Trust | |
| Chase Farm Hospital | Royal Free London NHS Foundation Trust | Antenatal service only |
| Ealing Hospital | London North West Healthcare NHS Trust | Antenatal service only |
| Guy’s Hospital | Guy’s and St Thomas’ NHS Foundation Trust | |
| King George Hospital | Barking, Havering and Redbridge University Hospitals NHS Trust | Antenatal service only |
| St Bartholomew's Hospital | Barts Health NHS Trust |
9.4 Predictive testing in pregnant women with threatened preterm labour: A best practice toolkit
London Maternity Clinical Network
Predictive testing in pregnant women with threatened preterm labour: A best practice toolkit
The aim of the toolkit is to optimise the understanding and uptake of fetal fibronectin (fFN) testing in pregnant women with threatened preterm labour (TPTL) in London. The toolkit presents the principles underlying the use of predictive testing rather than an exhaustive guideline. The Maternity Clinical Network recommends that all maternity units develop preterm labour clinical guidelines in line with these principles.
Current management of threatened preterm labour
Birth before 34 completed weeks of pregnancy is a significant cause of perinatal mortality and morbidity in the UK.
Current interventions that would be considered in a pregnant woman at high risk of imminent delivery of a premature baby include admission to hospital for monitoring, administration of steroids or tocolytic drugs to the pregnant woman and possible transfer to a specialist unit with appropriate staffing and expertise to most successfully care for a baby born at the extremes of viability.
NICE guidance on preterm labour and birth recommends treating all pregnant women in TPTL prior to 30 weeks5. This means most pregnant women in TPTL are likely to be treated. However, most pregnant women presenting with symptoms of TPTL (contractions without cervical change) will not deliver within seven days and more than 50% will eventually deliver at term. Treating all pregnant women in TPTL prior to 30 weeks results in;
- Unnecessary admission to maternity services.
- Unnecessary exposure of the baby to steroids.
- Unnecessary transfer to other units with the required level of neonatal care.
All of these incur risk or inconvenience to the pregnant woman and baby, as well as considerable financial costs to the NHS. Predictive testing could therefore help to reduce inappropriate intervention in pregnant women with TPTL.
Tests to predict imminent delivery in threatened preterm labour
Table 1 shows the most common predictive tests for threatened preterm labour. Each test has an individual ratio prediction of preterm birth less than seven days from presentation.
New educational tool – QUiPP app
QUiPP app is a clinical decision-making tool which can be used to calculate birth prediction for pregnant women with symptoms of threatened preterm labour as well as asymptomatic high-risk pregnant women.
The application has been designed for health, allied health and health research professionals who look after pregnant women to calculate individualised percentage risks scores of delivery within pre-specified clinically relevant timeframes. It is designed to be used with pregnant women as an educational tool and to arrive at shared decisions regarding the management of their pregnancy.
It relies on a relevant clinical history having been taken regarding the pregnant woman’s risk factors for preterm birth and her current symptoms. It relies on existing point-of-care testing: quantitative fetal fibronectin (fFN) sampling of the cervico-vaginal fluid and/or transvaginal ultrasound cervical length measurements.
The app enables clinicians to make clinical decisions based on numerous factors. For most episodes of TPTL, risk of delivery within seven days will be less than 1%. Precise thresholds for intervention will depend on the clinical setting, but it is recommended that a risk of delivery of delivery of less than 5% in the next week may justify withholding antenatal steroid administration, admission or in utero transfer.
Best practice
Transvaginal ultrasound cervical length and quantitative fFN alone or on combination are likely to be the most useful predictive tests for the management of TPTL, particularly when combined with other risk factor scores gained from using QUiPP app. Intervention is only likely to be justified where the risks of delivery within seven days is greater than 5% or where other tests are used (such as PartoSure™ or Actim® Partus) that are positive. Decisions will be tailored to individual circumstances, but any interventions for lower risk pregnant women require discussion with the obstetric consultant on-call and neonatal colleagues. Acting appropriately results in a number of potential benefits:
Pathway efficiencies resulting from:- Reduction in hospital admissions.
- Reduction in in utero transfer rate (ambulance journeys).
- Reduction in planning and administrative time for arranging an in utero transfer.
- Reduction in drug use, such as antenatal steroids.
Improved maternal experience by:
- Avoiding unnecessary hospital admission Avoiding ambulance transfer to an unfamiliar maternity unit.
- Providing reassurance that perterm delivery is not imminent.
Increased capacity by:
- Reducing the number of beds occupied by unnecessary admission and transfer. Currently, 47 per cent referrals to the Emergency Bed Service in London for in utero transfer are unsuccessful because there is no capacity; the median administrative time spent on these failed transfers is 340 minutes, involving discussions with between six and eight units.
Principles for the use of predictive testing in London
- All providers will have access to transvaginal ultrasound cervical length and/or quantitative fFN.
- Transvaginal ultrasound cervical length measurements and/or quantitative fFN are recommended in the management of pregnant women with threatened preterm labour with intact membranes between 22 and 35 weeks’ gestation.
- A fibronectin swab will be used to sample cervico-vaginal fluid in the posterior fornix in pregnant women with intact membranes and cervical dilatation < 3cms. The swab should be taken prior to digital examination.
- Recent sexual intercourse (within 48 hours) may falsely elevate quantitative fFN, but a low test result may still be relied upon.
- All providers will have access to QUiPP app.
- If the QUiPP app indicates risks the risk of delivery within seven days is less than 5%, steroids, admission and transfer may be withheld.
- Providers will have local guidelines for the use of predictive testing. In light of NICE guidance, these tests should include rationale for use of tests such as such as PartoSure™ or Actim® Partus.
- The different predictive tests should be used as indicated by the manufacturer.
- In utero transfer because of perceived risk of preterm labour (in pregnant women with intact membranes) should not occur without prior predictive testing and ideally combined with the use of QUiPP app.
Importance of education
The benefits associated with predictive tests and QUiPP app will not be realised if clinicians do not perform and interpret results correctly. Implementation is more extensive than purchasing swabs and point of care testing equipment. It is therefore essential that all units have up-to-date preterm labour clinical guidelines which include information on risk assessment and detailed protocols on the predictive test(s) used within the unit and interpretation of findings.
Auditable standards for threatened preterm labour
The Maternity Clinical Network encourages providers to carry out regular clinical audit on TPTL. Auditable standards include:
- The number of pregnant women presenting with TPTL who had a predictive test, alone or combined with QUiPP app tool, undertaken on arrival
- The number of pregnant women transferred to your hospital in TPTL who had a predictive test, alone or combined with QUiPP app tool, undertaken on arrival
- The number of pregnant women with TPTL producing a negative predictive test result (and QUiPP app risk of delivery that did not meet the thresholds) who went on to receive steroids, tocolysis, were admitted to hospital and/or were transferred to another unit.
- The number of pregnant women with TPTL and a positive predictive test result and QUiPP app risk of delivery that did meet the thresholds who went on to receive steroids, tocolysis, were admitted to hospital and/or were transferred to another unit.
Table 1: Available predictive tests
| Predictive Test | Threshold | Prediction of preterm birth < 7 days from presentation | Evidence Summary | NICE 2015 | Comments | |
| PLR | NLR | |||||
| Cervical Length | <15mmContinuous variable** | 4.32-13.64 | 0.03-0.77 | Broad evidence base mostly confirming moderately useful prediction | 1st line (> 30 weeks) | Sonographic training limits applicability in emergency setting |
| Actim® Partus(PHIGFBP-1) | 30 microg/L | 2.16-12.27 | 0.07-0.41 | Limited evidence, largely low positive prediction, rejected by NICE | Rejected | Some utility as rule-out test |
| Qualitative fetal fibronectin (fFn) | 50ng/mL | 2.73-8.9 | 0.09- 0.44 | Broad evidence base, mostly confirming moderately useful prediction | 2nd line (>30 weeks) | Relatively expensive |
| Quantitative fFN | 200ng/mL Continuous variable** | 9.69 | 0.44 | Growing body of evidence for predictive utility across risk range | Not examined | More targeted prediction may justify expense. Only test with useful long-term predictive value also |
| PartoSure™ (placental alpha-microglobulin-1) | 4 pg/microl | 16 | 0.21 | Minimal data (2 papers, <400 women in total) | Not examined | Awaiting robust evidence |
*Positive Likelihood Ratio
The positive likelihood ratio reports the number of times more likely pregnant women in preterm labour are to have that symptom than pregnant women not in preterm labour. A value between 5 and 10 is regarded as moderately useful.
*Negative Likelihood Ratio
A value of 0.1 and 0.2 is moderately useful and a NLR close to 1demonstrates that a negative test is equally likely for pregnant women in preterm labour and those who are not.
Where tests provide continuous variables, combining them with other risk factors in a predictive algorithm like QUiPP allows more tailored and accurate risk prediction than a single threshold.
Further reading:
- Abbott, DS, Radford SK, Seed PT, Tribe RM and Shennan AH. 2013. Evaluation of a Quantitative Fetal Fibronectin Test for Spontaneous Preterm Birth in Symptomatic Women. American Journal of Obstetrics and Gynaecology 208(2): 122-el.
- Deshpande SN, van Asselt ADI, Tomini F, Armstrong N, Allen A, Noake C, Khan K, Severens JL, Kleijnen I & Westwood ME. 2013. Rapid fetal fibronectin testing to predict preterm birth in women with symptoms of premature labour: a systematic review and cost analysis. National Institute for Health Research Health Technology assessment. 17(40).
- Fenton A, Peebles D & Ahluwalla J. 2008. Management of acute in-utero transfers: a framework for practice. British Association of Perinatal Medicine. 25th June 2008.
- Gale C, Hay A, Philipp C, Khan R, Santhakumaran S and Ratnavel N. 2012 In Utero transfer is too difficult: Results from a prospective study. Early human Development.88: 147-150
- Iams JD, Newman RB, Thom EA, Goldenberg RL, Mueller-Heubach E, Moawad A, Sibai BM, Caritis SN, Miodovnik M & Paul RH. 2002. Frequency of Uterine Contractions and the Risk of Spontaneous Preterm Delivery. New England Journal of Medicine. 346(4):250-255.
- Kuhrt K, Smout E, Hezelgrave N, Seed PT, Carter J & Shennan AH. 2016. Development and validation of a tool incorporating cervical length and quantitative fetal fibronectin to predict spontaneous preterm birth in asymptomatic high-risk women. Ultrasound Obstet Gynecol. 47: 104–109.
- Leitich H, Egarter C, Kaider A, Hohlagschwandtner M, Berghammer P & Husslein P. 1999. Cervicovaginal fetal fibronectin as a marker for preterm delivery: A meta-analysis. American Journal of Obstetrics and Gynaecology. 180:1169-1176.
- Lotfi G, Faraz S, Al Swalhee N, Nasir R, Somini S, Abdeldayem R, Koratkar R & Ammar A. 2015. Evaluation Of Pamg-1 for the prediction Of preterm birth In patients symptomatic Of preterm labor. J. Perinat. Med. 43.
- National Collaborating Centre for Women's and Children's Health (UK). 2015. Preterm Labour and Birth. National Institute for Health and Care Excellence: Clinical Guidelines, London.
- Nikolova T, Bayev O, Nikolova N & Di Renzo GC. 2014. Evaluation of a Novel Placental Alpha Microglobulin-1 (PAMG-1) Test to Predict Spontaneous Preterm Delivery. Journal of Perinatal Medicine. 42(4).
- Perales-Puchalt A, Spinelli M, Diago V & Perales A. 2013. The negative predictive value of cervical interleukin-6 for the risk assessment of preterm birth. The Journal of Maternal-Fetal & Neonatal Medicine. 26(13):1278-1281.
- Porcellato L, Masson G, O'Mahony F, Jenkinson S, Vanner T, Cheshire K & Perkins E. 2015. ‘It's Something You Have to Put Up With’—service Users’ Experiences of in Utero Transfer: A Qualitative Study. BJOG : an international journal of obstetrics and gynaecology .122(13).
- Stutchfield PR, Whitaker R, Gliddon AE, Hobson L, Kotecha S & Doull IJM. 2013. Behavioural, Educational and Respiratory Outcomes of Antenatal Betamethasone for Term Caesarean Section (ASTECS Trial). Archives of Disease in Childhood-Fetal and Neonatal. 98(3):F195-F200.
- Tsoi E, Akmal S, Geerts L, Jeffery B & Nicolaides KH. 2006. Sonographic measurement of cervical length and fetal fibronectin testing in threatened preterm labour. Ultrasound in Obstetrics & Gynaecology. 27:368-3723
- Watson HA, Carter J, Seed PT, Tribe RM, Shennan AH. 2017. The QUIPP app: a safe alternative to a treat-all strategy for threatened preterm labour. Ultrasound in Obstetrics and Gynecology. 50(3): 342-346.
9.5 In utero transfer record

For enquiries regarding your hospital account, please contact your hospital Admin Team.
For all other enquiries, you can contact our development team at ahmed.zaima@AMZingSoftware.com
Log In
Register
Profile
Search for hospital details
Staff Entitlements
| First Name | Surname | Update Status | Admin | Arrange Transfer | New |
|---|
Hospital Details
Arrange a Transfer
Ex-Utero Transfer
+
weeks and days
Yes
No
Yes
No
- Cervical Length: mm
- Actim® Partus:
- Positive
- Negative
- Fetal fibronectin
- Qualitative
- Positive
- Negative
- Quantitative: ng/mL
- Qualitative
- QUIPP App risk in 7 days: %
- PartoSure™
- Positive
- Negative
Yes
No
Yes
No

 Map Key
Map Key


 Level 1
Level 1

 Level 2
Level 2

 Level 3
Level 3